
XeF4Xenon tetrafluoridesp3d2 hybridizationStructureShapeBond angleLone pairsAdiChemistry
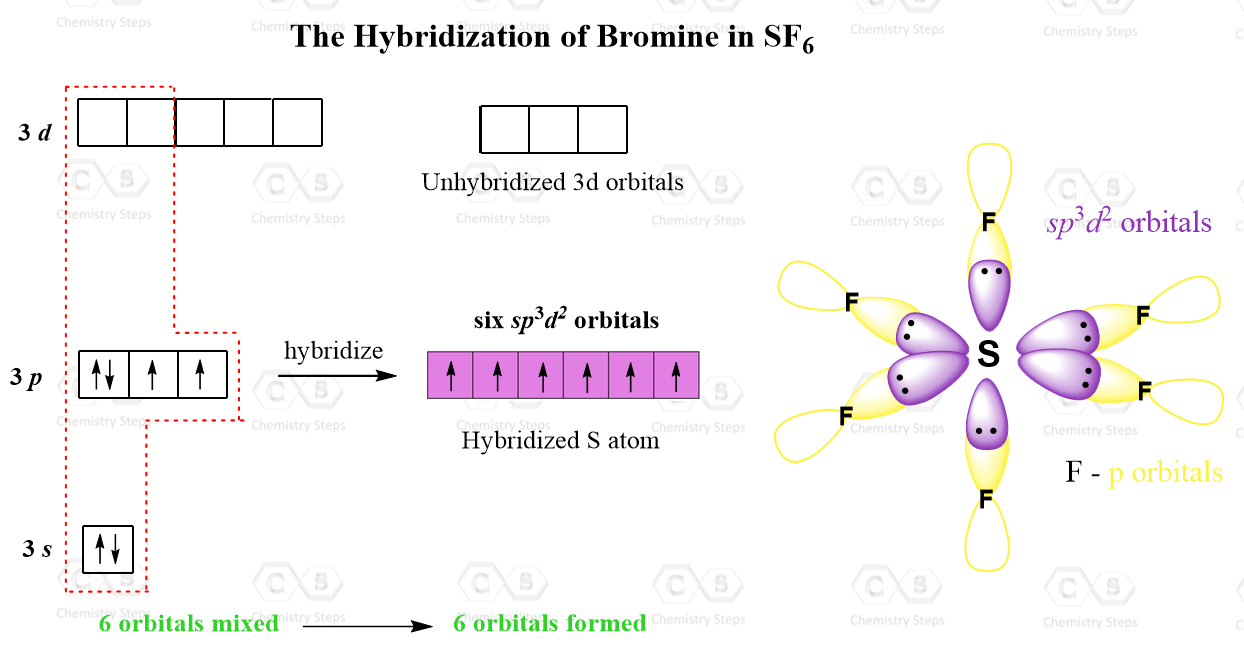
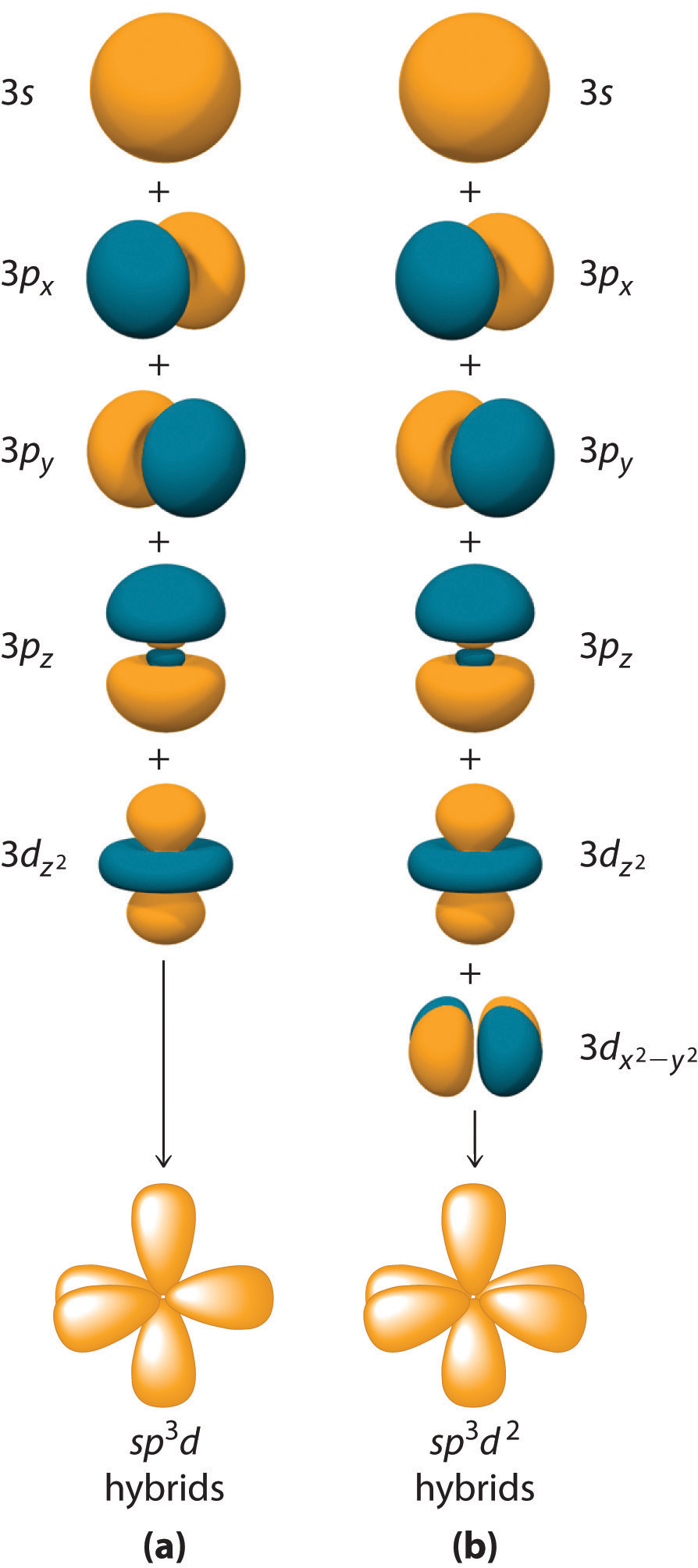
The sulfur atom in sulfur hexafluoride, SF 6, exhibits sp3d 2 hybridization. A molecule of sulfur hexafluoride has six bonding pairs of electrons connecting six fluorine atoms to a single sulfur atom. There are no lone pairs of electrons on the central atom. To bond six fluorine atoms, the 3 s orbital, the three 3 p orbitals, and two of the 3 d.

Hybridization Definition, Types, Rules, Examples
sp hybridization examples (Beryllium chloride, BeCl 2; Acetylene, C 2 H 2) sp 2 (Boron trichoride, BCl 3; Ethylene, C 2 H 4) sp 3 (Methane, CH 4; Ethane, C 2 H 6) sp 3 d (phosphorus pentachloride, PCl 5) sp 3 d 2 (sulfur hexafluoride, SF 6) sp 3 d 3 (Iodine heptafluoride, IF 7)

Sp3D2 Ujian
In this lecture discuss about the Sp3d2 hybridization will all examples. Sp3d2 Hybridization sigma and pi bond. Sp3d2 hybridization in SF6, XeF4, IF5 Gemotry.
sp, sp2, sp3, sp3d, and sp3d2 Hybridization Practice Problems Chemistry Steps
0:00 / 5:47 Part 5: sp3d Hybridization with Examples (Animation) Dr. Puspendra Classes 321K subscribers Subscribe Subscribed 592 views 1 year ago Hybridization - An Edu Series Complete.

PPT Lecture 11 Covalent Bonding Pt 3 Hybridization (Ch. 9.59.13) PowerPoint Presentation
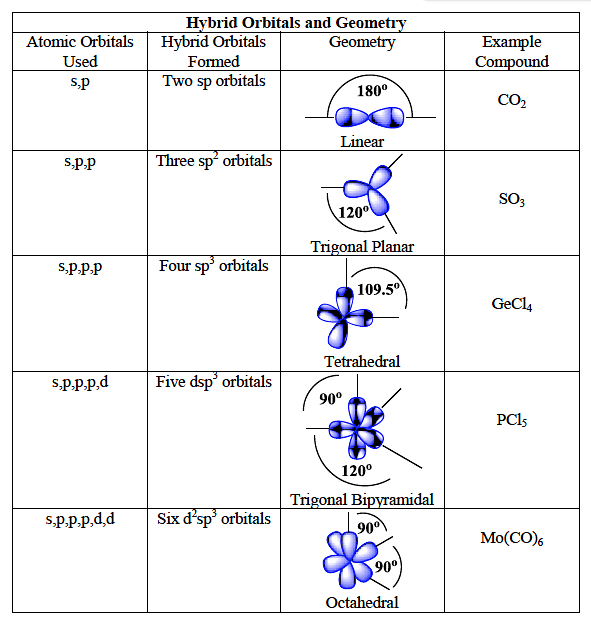
sp Hybridization. The beryllium atom in a gaseous BeCl 2 molecule is an example of a central atom with no lone pairs of electrons in a linear arrangement of three atoms. There are two regions of valence electron density in the BeCl 2 molecule that correspond to the two covalent Be-Cl bonds. To accommodate these two electron domains, two of the Be atom's four valence orbitals will mix to.
What is Hybridization? sp3, sp2, Examples and Formula
Once again, a linear geometry with a bond angles of 180 degrees. Let's do one more example using steric number to analyze the molecule. Let's do carbon dioxide. If we wanted to figure out the hybridization of the carbon there. Let's go ahead and do that. Using steric number. The hybridization of this carbon.
Hybridization sp, sp2, sp3 & sp3d Atomic Orbitals, Properties & Examples
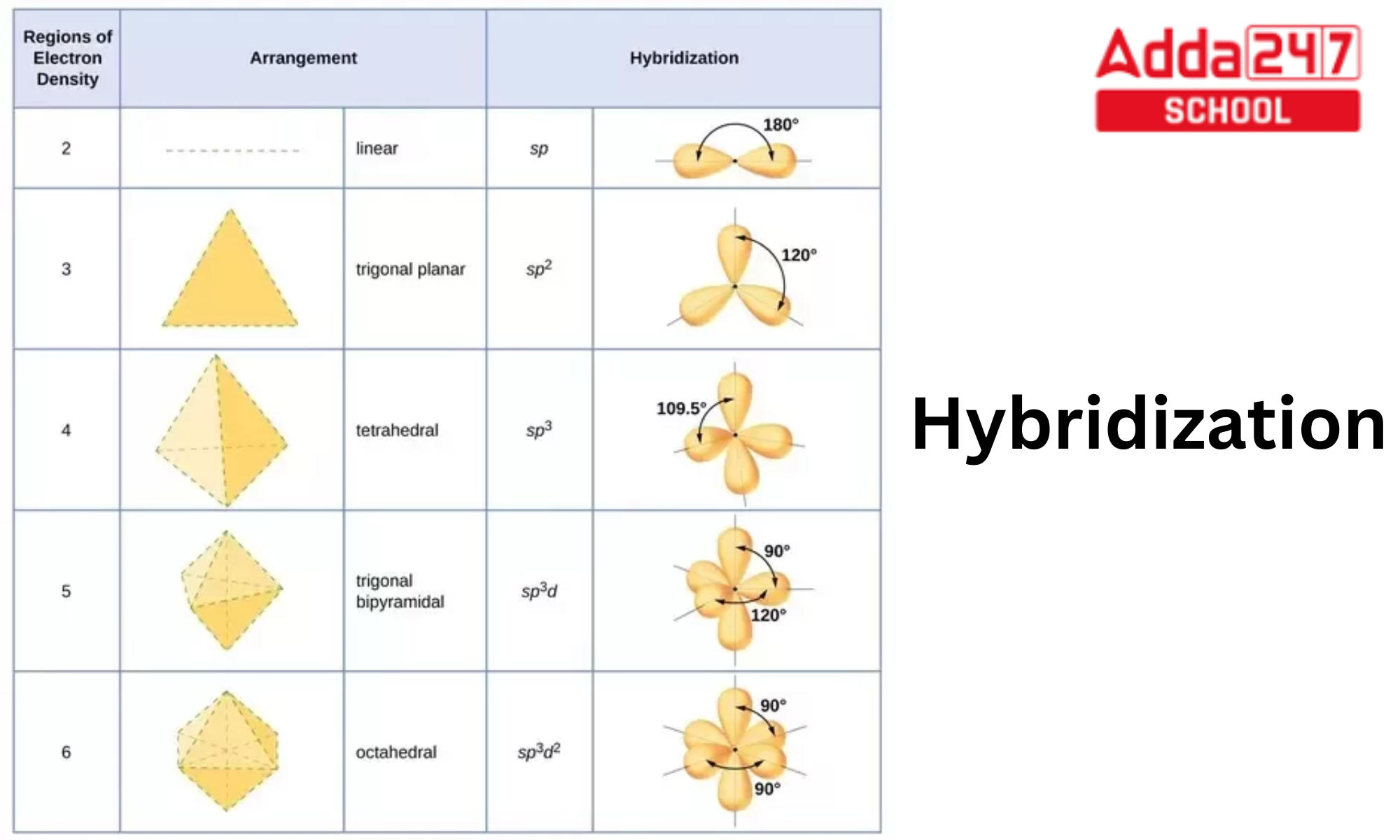
Valence bond theory: Introduction; Hybridization; Types of hybridization; sp, sp 2, sp 3, sp 3 d, sp 3 d 2, sp 3 d 3; VALENCE BOND THEORY (VBT) & HYBRIDIZATION. The valence bond theory was proposed by Heitler and London to explain the formation of covalent bond quantitatively using quantum mechanics. Later on, Linus Pauling improved this theory by introducing the concept of hybridization.

Hybridization Definition, Types, Rules, Examples
Steps to Find Hybridization The chemical process involves the reaction between two or more than two atoms. In chemistry, hybridization is the idea of combining two atomic orbitals to create a brand-new category of hybridized orbitals. Typically, this mixing creates hybrid orbitals with completely distinct energies, morphologies, etc. Hybridization
What is sphybridization? sphybridization kya hay? JEE/NEET/ IITJAM/ BSC Nursing
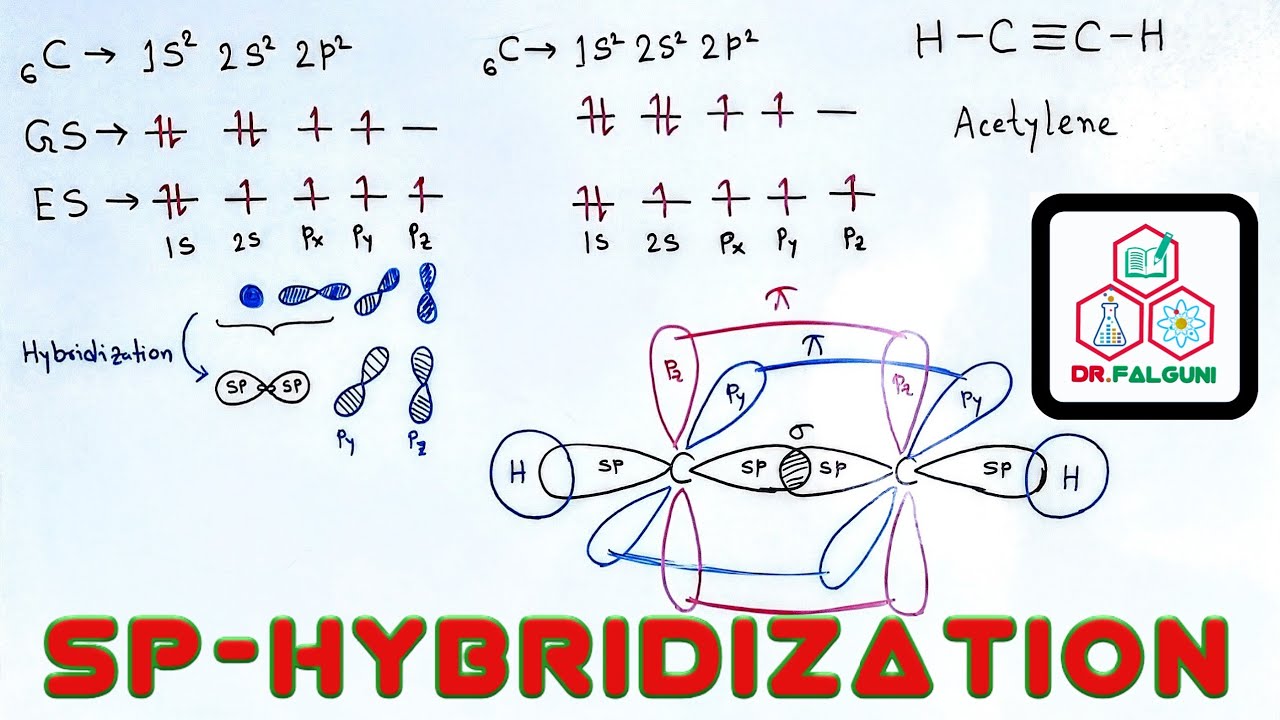
sp hybridisation, usually known as diagonal hybridisation, occurs when two s & one p orbitals belonging to the same primary shell of an atom combine to generate two new identical hybrid orbitals. The molecule generated due to this hybridisation has a linear shape with an angle of 180 degrees.
Localized Bonding and Hybrid Atomic Orbitals
Instead of three p orbitals and one s orbital, there are four sp 3 hybrid orbitals. Hybrid orbitals are extremely useful for explaining the characteristics of bonds, as well as predicting the geometry of different molecules. The latter makes use of VSEPR theory (Valence Shell Electron Pair Repulsion).

sp2 sp3 sp3d sp3d2 sp3d3 hybridization examples YouTube
July 30, 2019 sp 3 d Hybridization sp 3 d hybridization is shown in phosphorus penta chloride (PCl 5 ). The grounds state and the excited state outer electronic configurations of phosphorus (Z=15) are represented below.

Sp3d2 Hybridization with all examples/Chemistry with M.Faisal YouTube
Hybridization of s and p Orbitals. In BeH 2, we can generate two equivalent orbitals by combining the 2s orbital of beryllium and any one of the three degenerate 2p orbitals. By taking the sum and the difference of Be 2s and 2p z atomic orbitals, for example, we produce two new orbitals with major and minor lobes oriented along the z-axes, as shown in Figure \(\PageIndex{0}\).

Sp3d Orbitals
There are no lone pairs of electrons on the central atom. To bond six fluorine atoms, the 3 s orbital, the three 3 p orbitals, and two of the 3 d orbitals form six equivalent sp3d2 hybrid orbitals, each directed toward a different corner of an octahedron. Other atoms that exhibit sp3d2 hybridization include the phosphorus atom in PCl6−,PCl6−,

explain sp3d2 hybridization with an example Brainly.in
Trigonal hybridization in carbon: the double bond. Carbon and hydrogen can also form a compound ethylene (ethene) in which each carbon atom is linked to only three other atoms. Here, we can regard carbon as being trivalent. We can explain this trivalence by supposing that the orbital hybridization in carbon is in this case not sp 3, but is sp 2 instead; in other words, only two of the three.

Hybridization sp3d2 examples YouTube
sp 3 d2 Hybridization FAQs What Is Hybridization? Redistribution of the energy of orbitals of individual atoms to give orbitals of equivalent energy happens when two atomic orbitals combine to form a hybrid orbital in a molecule. This process is called hybridization .
Sp2 Hybridization Shape
sp3d2 hybridization And Examples.What is sp3d2 to hybridization?What is the geometry of sp3d2 to?How many hybrid orbitals are in sp3d2?Sp3d3 hybridizationSp3.